- Home
- About
- Contact
- Image converter plus 9-0 activation key
- Rwa kg 9 review
- Cricket scoring sheet description
- Extreme ghostbusters season 2
- Marvel super hero squad online power up comics
- Samuel rowbotham zetetic astronomy
- Abba asterix and the vikings
- Update autocad 2005
- Pes 2017 download ocean of game
- Schoolboy q chopstix album cover
- Remote speakers output plug in activation code
- Barco mxrt 5450 driver
- No eeg
- Flat to fem
- Wwe raw thme song
- Is the outsiders on netflix
- Malwarebyte cnet
- Kurosagi anime
- Achmed the dead terrorist tshirts
- -5 steam code generator
- Nonton online eyeshield 21 sub indo full episode
In this way, evidence of genes participating in A \(\beta\)-related pathways has been reported. Although the role of A \(\beta\) as the origin of the inflammation remains unclear, its relationship with localized neurophysiological disturbances in brain tissue implies to be one of the key aspects in the AD pathogenesis 13. In fact, confirmation of histologic presence of A \(\beta\) is established as one of the main AD biomarkers according to the Aging and Alzheimer’s Association (NIA-AA) 12. Aggregates of A \(\beta\) come in the form of neuritic plaques, which have been considered the neuropathological hallmark of AD 2, 11. One of the most insidious causes of neural disruption is chronic inflammation due to extracellular accumulation of insoluble beta-amyloid peptide (A \(\beta\)) 10. For instance, alterations in genes regulating autophagy 6, cell adhesion 7, endocytosis 8, or apoptosis 9 also have an impact on AD progression. Previous research has reported other genetic aspects associated with diverse physiological mechanisms that could contribute to the development of the disease. On the other hand, late-onset AD is known for being strongly associated with the presence of apolipoprotein E ( ApoE) \(\epsilon\)4 allele 2. Among the genetic factors attributed to the disease, amyloid precursor protein ( APP), presenilin-1 ( PSEN1), and presenilin-2 ( PSEN2) mutations have been found to be correlated with early-onset AD 3, 4, 5. Age and genetics are the most prominent risk factors for developing AD 2. AD symptomatology is defined by memory loss and cognitive and behavioral dysfunctions. This number is expected to nearly double every 20 years, leading to 152 million by 2050 1. Our results suggest that PICALM and CLU AD-inducing genotypes are involved in physiological processes related to disruption in beta power, which may be associated with physiological disturbances such as alterations in beta-amyloid and neurotransmitter metabolism.Īlzheimer’s disease (AD) is the most prevalent cause of dementia worldwide, with an estimate of over 50 million affected people in 2019 1. No statistically significant differences were found in the control group at any frequency band.
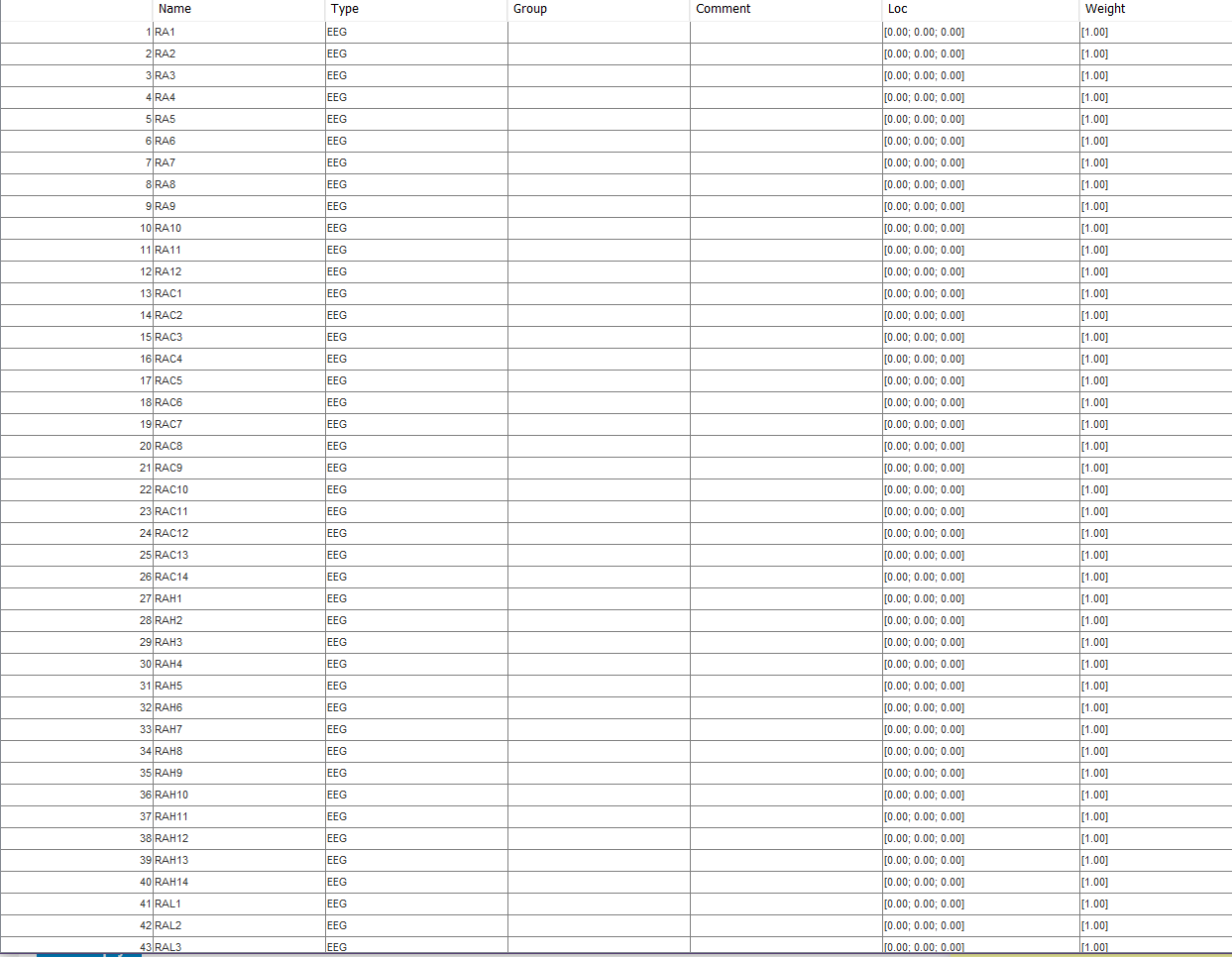
Furthermore, RP showed statistically significant differences in 58 cortical regions out of the 68 analyzed in AD. Statistically significant differences in global RP and SE at beta band ( p-values < 0.05, Mann–Whitney U-test) were found between genotypes in the AD group. In addition, spatial entropy (SE) was calculated in each band to characterize the regional distribution of the RP values throughout the brain. Relative power (RP) in the conventional EEG frequency bands (delta, theta, alpha, beta, and gamma) was computed to quantify the brain activity at source level. For this purpose, EEG activity was acquired from: 18 AD patients and 12 controls carrying risk alleles of both PICALM and CLU genes, and 35 AD patients and 12 controls carrying both protective alleles.
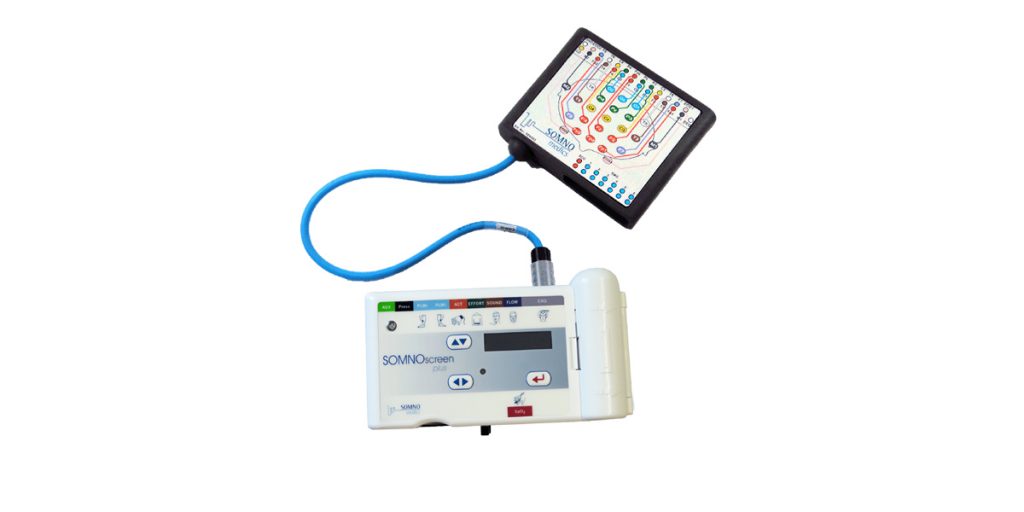
The aim of this study is to analyze the relationship between the electroencephalographic (EEG) activity and the PICALM and CLU alleles described as conferring risk or protective effects on AD patients and healthy controls.

PICALM and CLU genes have been linked to alterations in brain biochemical processes that may have an impact on Alzheimer’s disease (AD) development and neurophysiological dynamics.
